Decree 46/2026/ND-CP On Food Safety: What Businesses Must Do To Legally Announce Food Products
In practice, many food manufacturing and trading businesses in Vietnam face challenges when declaring products. Often, this is due to an inconsistent management system, which increases the risk of rejected dossiers or penalties.
The causes are not only internal operational issues but also the fact that Decree 46/2026/ND-CP introduces stricter requirements for food safety management. Businesses must now control the process continuously, from raw materials and production to distribution.
Consequently, the compliance focus has shifted from “meeting procedural requirements” to “ensuring actual operational capability.” So, according to Decree 46/2026/ND-CP, what should businesses prepare to declare food products correctly and minimize legal risks?
What Is Decree 46/2026/ND-CP And When Does It Take Effect?
Decree 46/2026/ND-CP is the legal document providing detailed guidance for implementing the Food Safety Law. It focuses on:
- Product declarations
- Food safety inspections
- Management of imported food
- Packaging and materials in contact with food
The decree was issued on January 26, 2026, replacing Decree 15/2018/ND-CP, which had been applied for many years. After a temporary suspension period until April 15, 2026, the decree officially takes effect on April 16, 2026.
Notably, the decree not only updates regulations but also changes the compliance assessment approach in practice. Businesses must now control their operations consistently, from documentation to actual practice.
Six Key Changes Businesses Must Understand
Decree 46/2026/ND-CP affects not only declaration procedures but also the entire food safety management system of businesses.
1. Narrowing self-declaration, expanding the scope of mandatory conformity declarations
According to Article 4, many product groups must now submit a conformity declaration instead of self-declaration. These include:
- Pre-packaged processed foods
- Food additives and processing aids
- Packaging and tools in direct contact with food
Risk: Incorrect declaration type or insufficient technical dossiers can lead to rejected applications, delaying market entry.
2. Stricter first-time management of packaging and tools in contact with food
Packaging and tools are now regulated separately:
- Must submit a conformity declaration
- Subject to state inspection on food safety
- Managed by the provincial People’s Committee regarding safety conditions
Risk: Poor control of input materials may result in sanctions or product recalls, even if the production process has no errors.
3. Mandatory implementation roadmap for HACCP and GMP
For the first time, Decree 46 requires high-risk product groups (e.g., health foods, food additives) to implement food safety management systems.
Businesses must complete the process by December 31, 2026, according to Articles 12, 15, and 54.
Risk: Superficial or delayed implementation may fail to meet actual inspection requirements.
4. Expanded and standardized procedures for imported food inspection
The decree expands inspection to include packaging and tools in contact with food.
Simultaneously, the inspection process is fully standardized:
- Defined inspection methods
- Required dossiers
- Sequence and processing deadlines
- Responsibilities of each stakeholder
Risk: Inconsistent or incorrect dossiers may require multiple resubmissions, delaying customs clearance.
5. Clear responsibilities for e-commerce platforms in food advertising
For the first time, Article 52, Clause 6, defines legal responsibilities of e-commerce platforms and intermediaries:
- Review and request sellers to provide legal documents
- Remove violating products upon request
- Bear joint liability if content is not properly controlled
Risk: Misleading or inconsistent advertising content may result in penalties, including online channels.
6. Regulations on timing and obligations for traceability
According to Articles 42 and 43:
- Businesses must store complete traceability data throughout the product lifecycle
- Immediate traceability is required when risks or warnings are detected
Risk: Failure to trace in time may prevent targeted recall, causing wider recalls, higher costs, and legal pressure.
Decree 46/2026/ND-CP not only updates regulations but also emphasizes actual control and demonstrable compliance. Businesses should prepare their operational systems, rather than merely completing paperwork.

6 key changes in Decree 46/2026/NĐ-CP that food businesses need to be aware of
Food Product Dossier And Declaration Process According To Decree 46/2026/ND-CP
1. Food Product Dossier
A basic dossier includes:
- Product declaration form
- Laboratory test results
- Business registration license
- Product label
- Food safety eligibility certificate
The key point is not the number of documents, but the consistency and traceability of the information. Even minor discrepancies in ingredients, parameters, or labeling can trigger requests for corrections.
2. Food Product Declaration Process
The process consists of five steps:
- Product classification
- Laboratory testing
- Preparing the dossier
- Submitting the dossier
- Receiving results
In practice, bottlenecks often occur not in the process itself but in uncoordinated preparation. This can lead to repeated submissions and prolonged processing time.

Documentation and dossiers for food registration under Decree 46/2026/NĐ-CP
What Businesses Should Do To Minimize Risks Under Decree 46/2026/ND-CP
To ensure effective compliance, businesses should adopt a systematic approach rather than handling each procedure separately.
Step 1: Review all existing dossiers
Compare documents such as the product declaration, test results, and labels. Correct discrepancies in ingredients, parameters, or units.
Step 2: Standardize operational procedures
Set up production, control, and data storage steps according to the declared content. This ensures information can be verified when required.
Step 3: Implement a food safety management system
Apply models such as HACCP or ISO 22000 to control hazards consistently. This also builds a data foundation for future audits.
Step 4: Establish a traceability system
Create a mechanism linking data from raw materials, production, and distribution. This helps quickly identify the affected scope in case of issues.
If the current system has not been reviewed according to Decree 46/2026/ND-CP, early assessment is essential. This is the only stage where businesses can proactively adjust before discrepancies impact operations.
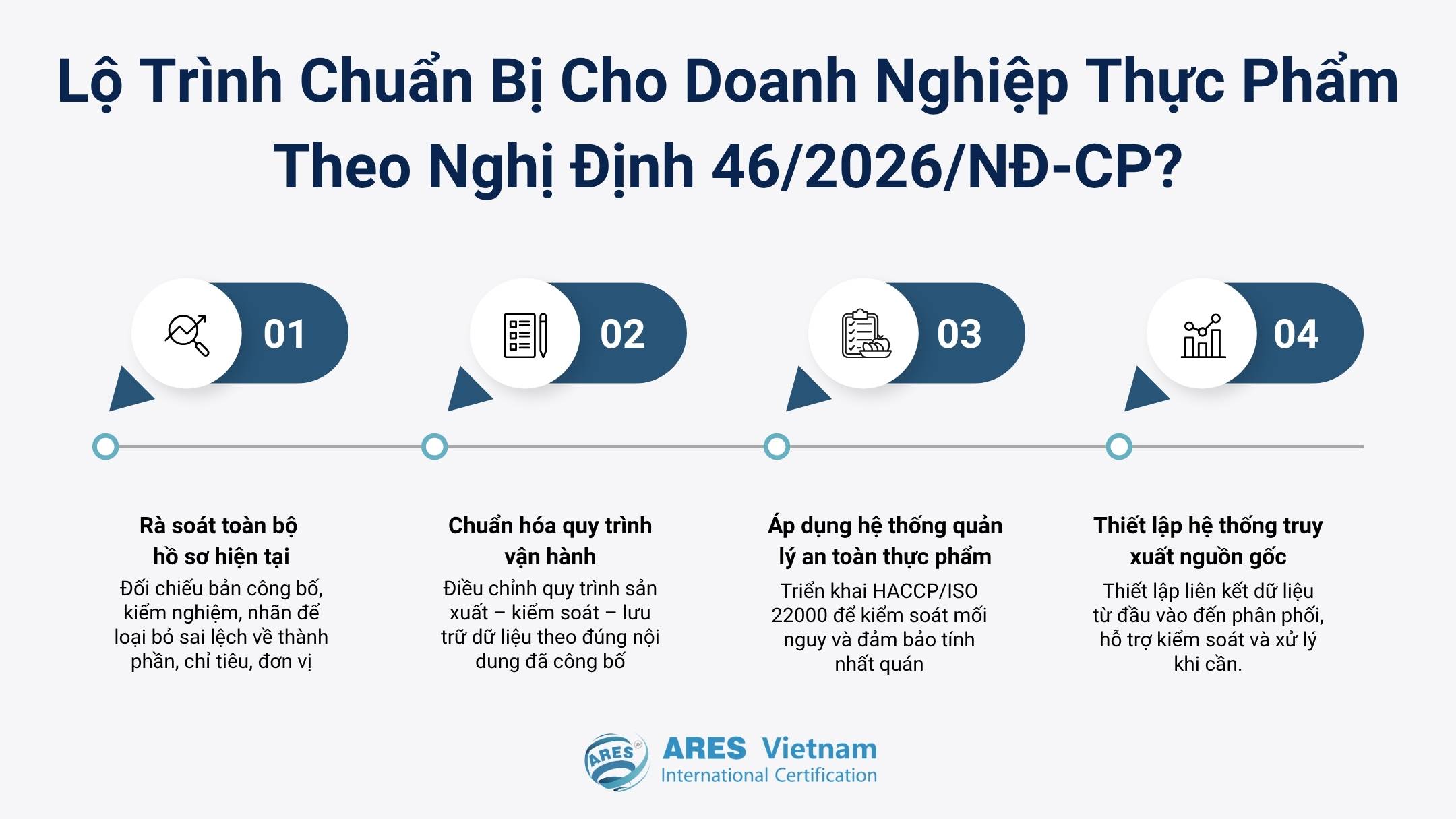
Preparation roadmap for food businesses under Decree 46/2026/NĐ-CP
ARES Vietnam’s Support Services For Food Businesses
In addition to providing reference information, ARES Vietnam supports businesses with ISO 22000 and HACCP certification services in line with Decree 46/2026/ND-CP.
Contact ARES Vietnam to have experts assist with:
- Assessing current compliance levels
- Identifying potential discrepancies
- Building systems aligned with actual operations
This helps businesses reduce risks from the start and ensures a smooth product declaration process.
Frequently Asked Questions About Food Product Declarations Under Decree 46/2026/ND-CP
Below are common questions businesses encounter when implementing food product declarations under Decree 46/2026:
| Common Questions | Answers to Inquiries |
| Which businesses does Decree 46/2026/ND-CP apply to? | It applies to domestic food manufacturing and trading facilities, importers, and entities involved with packaging or food contact materials. |
| Does Decree 46/2026 replace Decree 15/2018/ND-CP? | Yes. Decree 46/2026/ND-CP replaces and amends certain provisions of Decree 15/2018/ND-CP and introduces stricter food safety management requirements. |
| What documents are required for a food product declaration under Decree 46/2026? | A basic dossier includes the product declaration form, test results, business registration, product label, and other legal documents depending on the product type. |
| Why are food declarations often rejected even if all documents are prepared? | Rejections often occur because information between documents or between the dossier and actual production is inconsistent, failing verification checks. |
| Are businesses required to implement HACCP or ISO 22000? | Depending on the product group, especially high-risk products, businesses may be required to apply HACCP, GMP, or equivalent systems according to the prescribed roadmap. |
| What are the traceability requirements? | Businesses must maintain complete traceability data throughout the product lifecycle and provide it immediately upon request or in case of an incident. |
| What should businesses prepare before declaring a food product? | They should review dossier consistency, standardize operational procedures, and establish a traceability system to ensure verifiable compliance during inspections. |
Related news

ARES Vietnam Conducted ISO 14064 Certification Audit At SUN RISE BICYCLE CO., LTD
Read more


ARES Vietnam Conducted ISO 14064 – ISO 14067 Certification Audit At A&J Cambodia
Read more


ARES Vietnam Conducts ISO 9001:2015 Audit At VIETNAM CHO CHEN ACRYLIC CO., LTD
Read more


ARES Vietnam Conducts ISO 9001:2015 & ISO 14001:2015 Certification Audit At Electronic Technology YMX Vietnam Co., Ltd.
Read more


How Does ISO Help Upgrade Management In Vietnam’s Manufacturing Industry?
Read more














